
How to Calculate Density of Water Exampleįor example, if the mass is 9 g and the volume is 9 mL. Ensure to add the si unit to the final result which is in g/mL or gcm -3. Third Step (Solution): Now, you substitute your formula for the density of water with the data available to you and solve the problem. 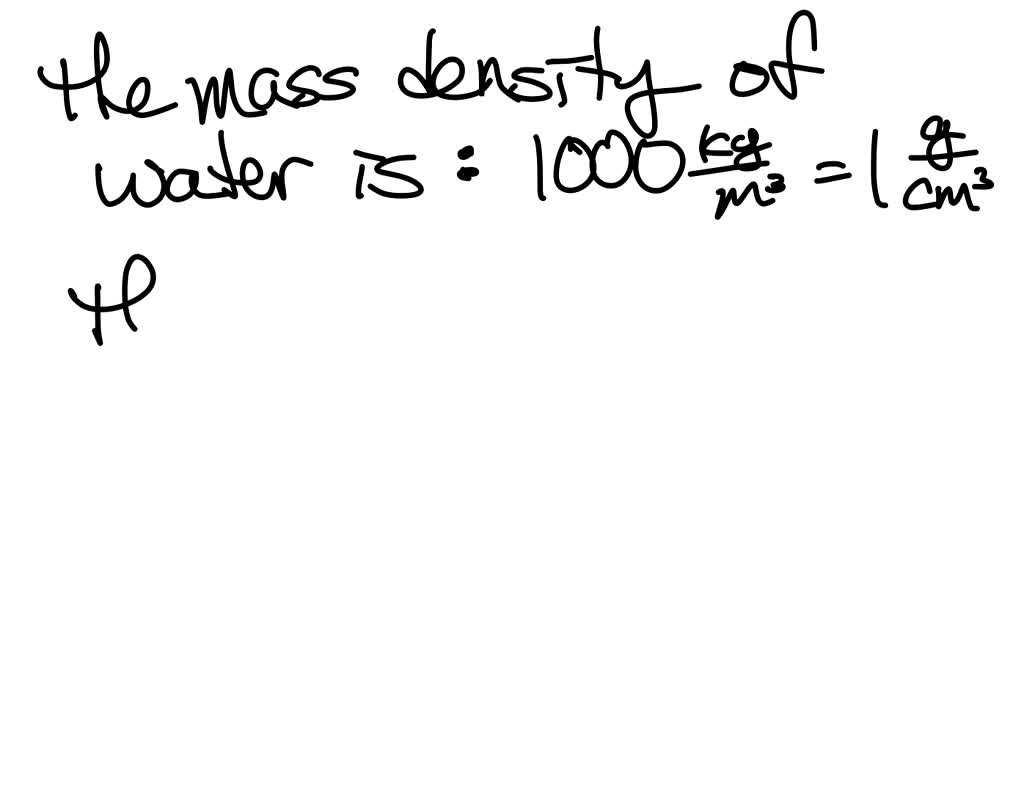
Second Step (Formula): In this stage, you state the equation that will help you to calculate the density of water, which is Density (ρ) = Mass (m) / Volume (v) or ρ=m/v. Here are three steps that will help us in calculating the density of water:įirst Step (Data): It is important to read and understand the question so as to gather all the necessary information. How to Calculate Density of Water: Step-by-Step Guide Ensure consistent units, such as grams for mass and cubic centimetres for volume, or kilograms for mass and cubic meters for volume, to obtain the density in the appropriate units. Then, plug these values into the formula and perform the division to find the density. Measure the mass of the water sample using a scale and the volume using a graduated cylinder or similar tool. To calculate the density of water, you use the formula: The density of water is approximately 1 gram per cubic centimetre (g/cm³) or 1000 kilograms per cubic meter (kg/m³) at standard atmospheric pressure and temperature.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
